So2 molecular geometry and bond angle1/18/2024 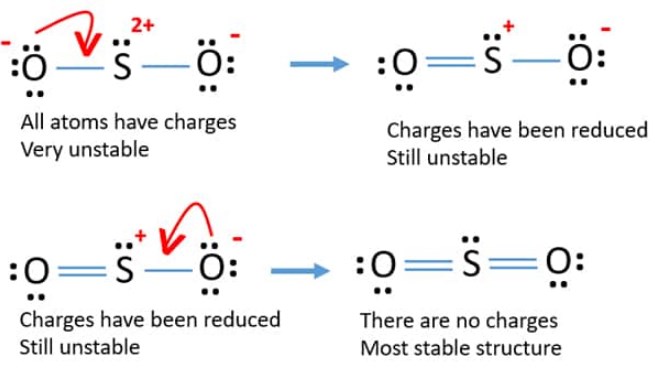 
This is because electrons are negative and they repel each other. First, we can use the Lewis structure for SO2 to visualize how the atoms and unbonded electron pairs will spread out in three dimensions. Without an only set, both the geometries coincide for any compound.We can find the molecular geometry for SO2 it two ways. At the same time, molecular geometry thinks about only atoms. So, electron geometry is different from molecular geometry because it thinks about all the electron sets (including lone sets) while determining the shape. We can quickly learn the molecular geometry of any substance utilizing the given chart. The molecular geometry of SO2 is curved, with a bond angle of 120 °. It causes a repulsion of electron pairs to create the 120-degree angle. One single atom of SulphurSulphur is bonded with two atoms of Oxygen covalently. There’s a picture connected below for a much better understanding. one of the 3p orbital and another electron in 3d) formed the bonds between sulphur and Oxygen. Thinking of the other 2 electrons of 3p, which were not related to hybridization? Et cetera, two paired orbitals extend the lone set of sulphur. Now, the 3s2 and 3p3 connect to create Sp2 hybridization with three comparable orbitals, including two paired electrons and two unpaired.įor developing two sigma bonds with oxygen atoms, sulphur requires the two unpaired electrons from the Sp2 hybridized orbitals. 
When in a thrilling state, one electron from 3px moves to a 3d orbital. Too, in case of SO2, the ground state electronic arrangement is 1s2 2s2 2p6 3s2 3p4. You can opt for the procedure.Ī fast side for you, when 1 s orbital unites with 2 p orbitals, obtains in Sp2 hybridization having three comparable orbitals. I would undoubtedly suggest recognizing the concept first and afterwards. Now can determine the hybridization of SO2 in 2 ways, one is the theory, and the second is the straight applying the formula. Oxygen has two single pairs, and SulphurSulphur has one lone pair.Īt last, do not forget to confirm the legal cost of all the atoms! SO2 Hybridization Last but not least, this will complete the octet of the atoms. We need to place these staying electrons around the atoms based on the demand. Subtracting that from the complete valence electrons, we obtain 10 electrons remaining. Hence the number of electrons utilized in double bonds = 8. There are two atoms of oxygen in the compound, thus = 6 * 2 = 12.Īfter drawing the structure, we can see that none of the atoms can accomplish their octet with single bonds. In SO2, the Sulphur’sSulphur’s valence electron = 6.Īnd also, the valence electrons of oxygen = 6. Now allow’s see the lewis structure of SO2. The comprehensive collection of bonding electrons around the boron is 8 (complete octet). The variety of valence electrons for boron is 3. so2 hybridization: For example, using this to BH4, we get. This formula explicitly shows the connection between the variety of bonding electrons and their web link to the number officially “kept” by the atom. 
Official cost computation can be done utilizing:. After that, could you bring them to the electropositive ones?ĥ– Providing double or triple bonds is fundamental if it is needed for satisfying the octet regulation for all atoms.Ħ– At last, it’s essential to examine if all the atoms have their cheapest possible official cost. Constantly start with the electronegative atoms. The atom with the highest possible number of binding places is the central atom.ģ– The third step is to develop a skeletal system framework with single bonds.Ĥ– Following, our job is to achieve the octet of the atoms with the resting electrons after forming the solitary bonds. 2– Next point is determining the central atom.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
